Prices’ spikes and declines; changes in distribution; record numbers of new registrations approved; lower costs for pesticide laboratory testing.
Flavio Hirata is an agronomist (Esalq/USP), MBA, specialized in pesticide registration and partner at AllierBrasil.
AllierBrasil is a pesticide registration consulting firm having approved 404 registrations, from 2006 to 2024, ranked # 1 in approvals in Brazil. In March AllierBrasil is organizing AgriTour China, with technical-commercial visits to factories and to CAC Shanghai 2026.
Brazil is the global agricultural leader, with a harvest forecast of 354.4 million tons of grains in the 2025/26 in more than 80 million hectares. The growing adoption of technologies as they become more accessible – whether due to price and/or availability – combined with other factors, has driven successive records in grain production and yields.
Grain production vs. area vs. yield in Brazil (2016/17–2025/26)
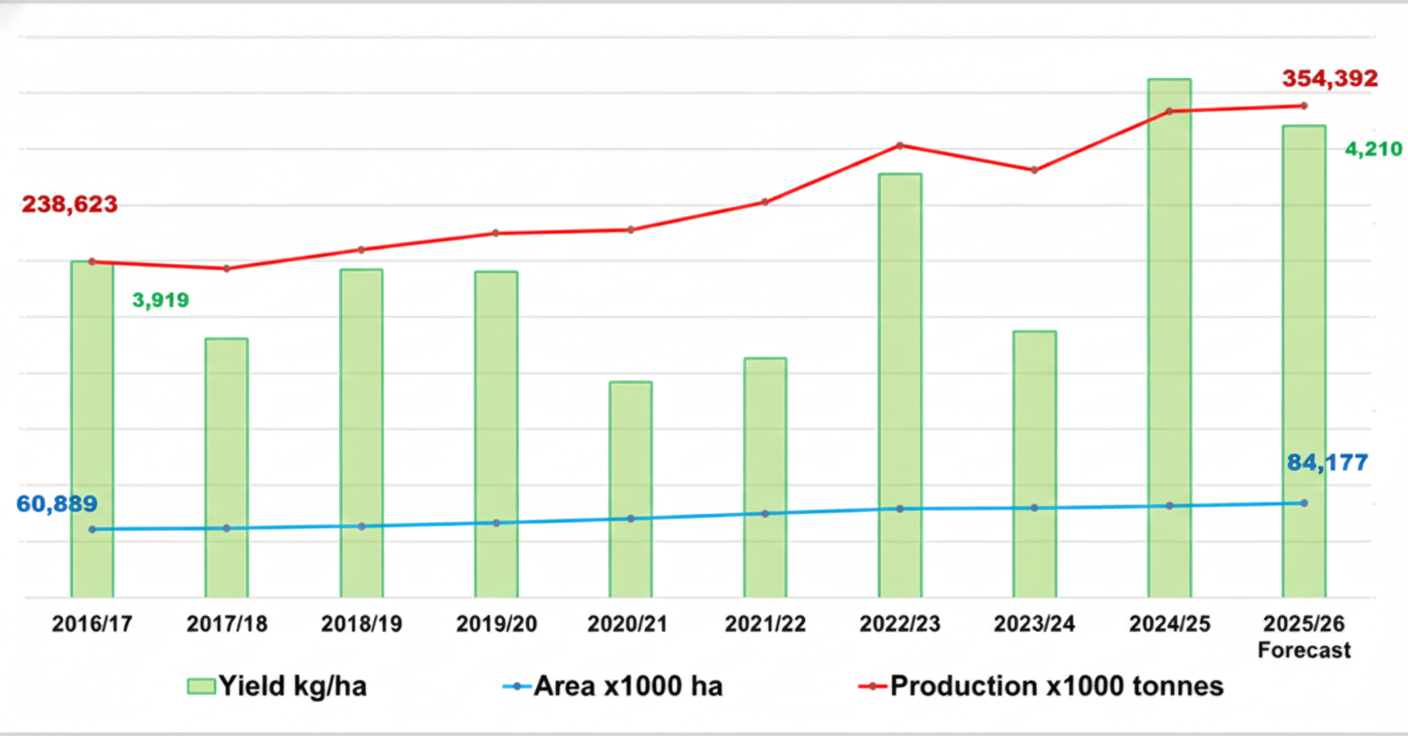
Source: National Supply Company (Conab); adapted by AllierBrasil
This context has intensified the interest of foreign companies and reshaped how local players operate over the years, especially in pesticides, essential for large-scale production. To give a sense of scale, in the early 2000s there were fewer than 20 distributors (companies holding pesticide registrations), whereas today there are more than 400 – particularly companies focused on generic (equivalent) chemicals and, more recently, biological products.
The interest in the pesticide market changed when Brazil regulated the registration of equivalent (generic) products, with the first approvals starting in 2006. This reduced the scope of laboratory testing required for registration, lowering costs from around US$ 500,000 to US$ 150,000, and shortening the approval timeline to 12–15 months.
Not everything was expedited, however. The average time to obtain the Temporary Special Registration (RET), which was required to import products and conduct laboratory testing, exceeded 24 months, which was nearly twice the time needed to approve a technical-grade active ingredient and its corresponding formulated product. For many years, the RET was a major bottleneck in the registration process.
From that point onward, there was a rush to enter the market, mainly by manufacturers from China and, to a lesser extent, from India (both manufacturers and traders), while local distributors also accelerated efforts to register products. As generic registrations were approved, prices decreased significantly and, over time, the reluctance to import and use products from China gradually diminished.
As more registration requests were submitted, the timelines began to lengthen, reaching as much as 10 to 12 years.
Foreign groups and investment funds started acquiring stakes in, or fully acquiring, local distributors with strong appetite. Given the long time required for approvals, a secondary market also emerged for trading existing product registrations.
Everything changed during the pandemic: supply shortages, extremely high ocean freight rates, and speculation led to product scarcity and sudden price increases – up to 400% in some cases – with 100% of payment upfront. High prices and strong demand encouraged Chinese manufacturers to expand production capacity.
After the pandemic, further changes followed: production capacity became far greater than demand, freight costs declined and local inventories were excessive, which is a strategy adopted by distributors to ensure product availability in the event of renewed shortages. This was followed by a steep collapse in prices, reaching levels never seen before. Manufacturers began selling at production costs, or even below, just to keep plants operating, since maintaining idle factories is extremely expensive. This was compounded by the appreciation of the Brazilian currency in relation to dollar. Some companies held inventories purchased at higher dollar prices, while new shipments were arriving with the same products priced lower in dollars.
With prices so low, exporters began offering distributors more generous credit, including longer payment terms, without the usual warranties. This tends not to create problems when the market is operating smoothly. However, the approach is fragile in the face of court-supervised reorganization proceedings (bankruptcy protection, similar to US’ Chapter 11), both in the number of companies involved and in the amounts at stake, which can be enormous, creating a domino effect across the entire distribution chain, as the market is experiencing today.
Meanwhile, the number of registration approvals has reached record levels in recent years, and this trend is expected to continue in 2025, potentially exceeding 900 new registrations. At the same time, delays in registration evaluations have driven companies to file lawsuits seeking analysis within the deadlines established by law. The number of such lawsuits has increased significantly over the past five years. Today, more than 20% of approvals of generic formulated products are granted in compliance with court decisions. In 2025, Anvisa had already approved 115 toxicological evaluations by court decision, and Ibama, 48 environmental evaluations (as of December 15th).
RET is no longer mandatory and is now required only for new active ingredients or new uses. Over the past two years, regulatory requirements have increased significantly, including SISPA, risk assessment, quality control, and other obligations. On the other hand, with more Good Laboratory Practice (GLP) laboratories operating in Brazil, the cost of a dossier for an equivalent formulated product has fallen from around US$ 75 thousand to about US$ 25 thousand, compared to the prices seen in the early 2000s.
Approved Pesticide Registrations (2021 – 15/12/2025)
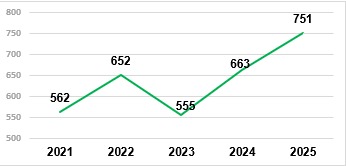
Source: Ministry of Agriculture (MAPA); adapted by AllierBrasil
Despite the challenges, Brazilian market continues to attract more companies. From abroad, this includes manufacturers with a limited portfolio, large formulators, and traders previously focused on China’s domestic market and Southeast Asia. In Brazil, global grain exporters have begun importing directly from manufacturers in China and India, and mid-sized distributors have started importing from China as well. The objective is to serve customers reducing intermediaries in the distribution chain and keep competitive margins, while assuming the risks associated with weaker links in the distribution structure.
To facilitate the search for Brazilian and international suppliers, the look2agro™ platform provides contact information for more than 80,000 companies from Brazil, China and India across multiple agribusiness segments.
To connect with suppliers in China and India, AllierBrasil is organizing the AgriTour China – CAC Shanghai in March 2026. The program includes visits and meetings with companies interested in the Brazilian market, as well as visit to CAC Shanghai, the world’s largest agrochemical trade fair.

